|
11/13/2023 0 Comments Element o atomic number Even the masses of protons, neutrons, and electrons are ratios of their mass to carbon. The mass of an atom of another element is the ratio of its mass to the mass of a carbon atom. The mass of a 12C carbon atom is specified as exactly 12. The only exception is carbon, whose mass was used as a standard reference. An amu is 1/12 the mass of one atom of 12C, or about 1.66 x 10-27 kg.Ītomic mass values for elements are almost never an integer. One atomic mass unit is a very small amount of mass. The mass of an atom or particle is expressed in atomic mass units or amus. To get an idea of how small an atom is, consider that a small gold coin may contain over 20,000,000,000,000,000,000,000 atoms. The smallest particle that would still have the properties associated with gold is an atom. How fine can the dust become and still be considered gold? The density of the cloud at any point is the probability of finding the electron at that point.įigure 10. The probability of finding the electron in any region of space can then be described by a cloud that rapidly thins out as one goes farther from the nucleus. We might think that the electron is rapidly moving around the nucleus and our experiment "catches" the electron as an instantaneous "snapshot" of it in motion. However, if we repeat this experiment many times, it will be found that the electron is much more likely to be located in certain regions of space surrounding the nucleus than in other regions of space. If we attempt to detect an electron in an atom, we might find evidence of it located almost anywhere around the nucleus. The electrons of the atom move rapidly around the nucleus. Because the protons are positively charged, the nucleus has a positive electric charge. In an atom, the protons and neutrons clump together in the center and are called the nucleus. 1999‑2023 - All Rights Reserved.If the proton and neutron were enlarged, and each had the approximate mass of a hippopotamus, the electron, enlarged to the same scale, would have less mass than an owl. Retrieved from Ĭopyright © Israel Science and Technology Directory. "Sortable list of elements of the Periodic Table". The story behind the discovery that elements are born in stars.Atomic Weights of the Elements (From IUPAC).Multilingual Dictionary and Etymology of the Periodic Table Elements.Atomic Reference Data for Electronic Structure Calculations.List of Periodic Table Elements in Hebrew.Other resources related to the Periodic Table For these elements, the weight value represents the mass number of the longest-lived isotope of the element.Įlectron configuration: See next page for explanation of electron configuration of atoms. The elements marked with an asterisk have no stable nuclides. The values shown here are based on the IUPAC Commission determinations ( Pure Appl. 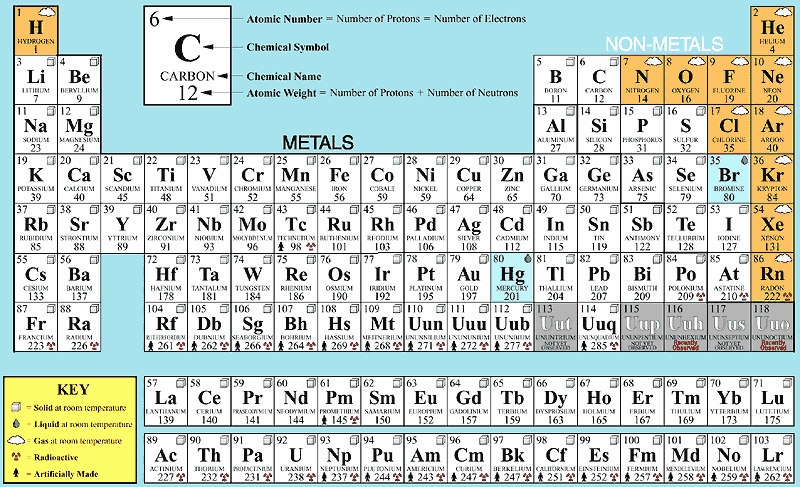
For relative abundances of isotopes in nature, see reference on Atomic Weights and Isotopic Compositions.Ītomic weight: Atomic weight values represent weighted average of the masses of all naturally occurring isotopes of an element. The abundance of each isotope depends on the source of materials. For example, the two common isotopes of carbon, 12C and 13C, have 6 and 7 neutrons, respectively. Elements have more than one isotope with varying numbers of neutrons. The isotope of an element is defined by the sum of the number of protons and neutrons in its nucleus. Isotope: Atoms of the same element with the same atomic number, but a different number of neutrons. Thus, each proton and neutron has a mass of about 1 amu. This isotope of carbon has 6 protons and 6 neutrons. Atomic mass is measured in Atomic Mass Units (amu), which are scaled relative to carbon, 12C, that is taken as a standard element with an atomic mass of 12. Each element is uniquely defined by its atomic number.Ītomic mass: The mass of an atom is primarily determined by the number of protons and neutrons in its nucleus. 
Boiling pointĪtomic number: The number of protons in an atom. For the Year of Discovery of elements see the list with the English and Hebrew names. 
For these elements, the weight value shown represents the mass number of the longest-lived isotope of the element. The elements marked with an asterisk (in the 2nd column) have no stable nuclides.Lanthanoids and Actinoids are numbered as 101 and 102 to separate them in sorting by group. Group: There are only 18 groups in the periodic table that constitute the columns of the table.Elemental compositions of crustal rocks differ between different localities ( see article). Earth crust composition average values are from a report by F.In a sorted list, these elements are shown before other elements that have boiling points >0☌. The density of elements with boiling points below 0☌ is given in g/l.List of Periodic Table elements sorted by → Atomic number No. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
